
Lab notebook
Nothing is as valuable as your raw data. If there is a fire in the lab, grab your notebook!
All the information at your desk should be accessible within 5 minutes. Time is too precious to waste looking for a method you saw in a paper last week or trying to find the results of last week’s experiments. Your lab notebook should be well organized and clearly legible for you to review, but more importantly, it should be easy to access for any other individual who might need to read or repeat your work.
The record of every experiment should contain:
- The date of the start of the experiment.
- The title of the experiment.
- A brief statement of purpose.
- The data collected in the experiment.
- A copy of a protocol should be pasted in and amended as you go.
- A record of calculations.
- Remember to record everything. You will not be able to remember everything. Omitting a result is falsifying data.
- At the end of the experiment, write a one-sentence summation of the experiment results. Note any oddity or aberration, and add comments about why the experiment may or may not have worked.
Academic laboratories tend to be much more liberal in their lab book requirements, and many have no rules or guidelines whatsoever. You may find everything from data kept on paper towels to bound notebooks of printer paper. However, once scientists move to industry, they often quickly realize scientific accountability and IP are significantly more important. Most companies will not, and should not, tolerate sloppiness in data-keeping.
Protocols & SOPs
Protocols and SOPs (Standard Operating Procedures) pursue two goals:
- To provide a clear step-by-step guide for all processes and activities. The document provides clear and detailed steps for repetitive tasks and processes. But it’s also used for the training of new staff and employees.
- To avoid any hazardous situation and dangerous accidents in the lab by pointing out the risks and explaining the correct procedures (such as handling chemicals, cells, and viruses of certain risk levels, avoiding exposure to UV light or high magnetic fields, etc.)
No matter how experimental your procedure is, every experiment must have a protocol—every time. Even if you are working a technique out for the first time or repeating a commonly used procedure, you should do it according to a written protocol you’ve prepared beforehand.
To start writing your protocols and SOPs:
- Think of the specific process in detail. Write down all the steps you need to follow while doing it.
- To check the clarity of the steps, go through them with a blind mind: Repeat the processes not as you know them, but as your protocol or SOP explains them to you. Find out if any detail is missing.
- Ask a colleague to perform the task using your list and shadow them as they go through the tasks. Check for any missing and hazy points and clarify them for your audience.
If it’s your first time following a protocol, follow it strictly. You must be able to reproduce the usual results before you can start to vary conditions. You must know you are measuring the effect of a variable, not of your technique.
Over time, change the protocol as you see fit. As you proceed through the experiment, note down any improvements to the protocol that would make sense. When evaluating your data, rethink the alterations and check with someone in the lab to see if they make sense.
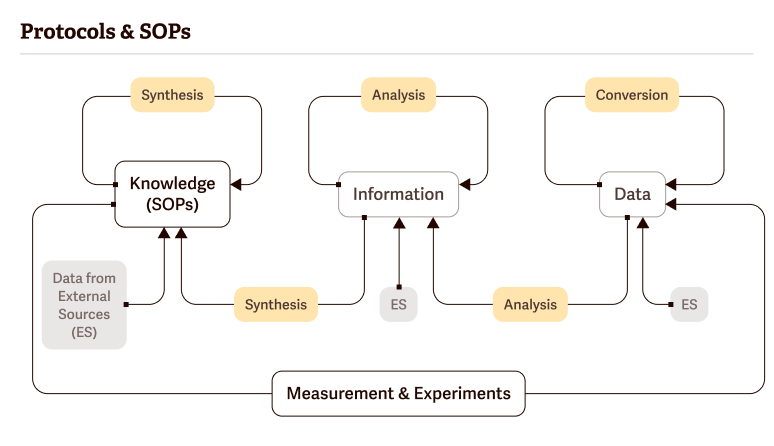
Keep in mind the following considerations:
- Prefer many simple steps over a few complicated steps. Nothing is too obvious.
- Make sure to include all risk variations.
- Avoid acronyms and non-standard unit systems.
- Review your protocols and SOPs every six months, in case there had been a reorganization of the lab or a change of the chemicals and solutions.
- Assign a specific team member to be responsible for each protocol, including regular reviews.
Choosing a Document Management System (DMS)
The vast majority of scientists still use paper notebooks in the workspace. They are cheap, easy to use, and they let you highlight notes and sketch figures in the margins. However, as scientists deal with ever-larger volumes of data (scientific data output doubles every three years), simply gluing results into a paper notebook and flipping through dusty pages becomes unsustainable.
Digital DMS’s allow scientists to:
- Minimize data loss by allowing users to retrieve data by user, tag, textual content, timestamp, and/or structured data queries;
- Reduce avoidable errors by enabling experiments to be shared, reviewed, and edited by colleagues in real-time; and
- Perform increasingly complex research by integrating with different lab equipment, data types, and file formats, so that scientists can bring together their data in a coherent fashion.
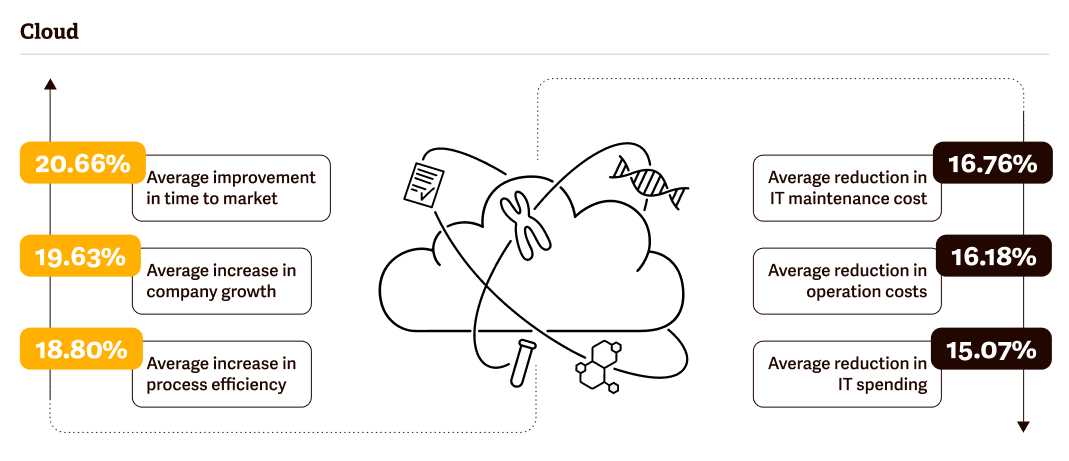
When compared to on-premise solutions (hosted on your local computers), cloud-based DMS’s then have a number of downstream benefits, including greater process efficiency, increased company growth, and faster time to market.
However, once you’ve chosen to go with a cloud-based DMS, there are still many other key differentiators you may want to look for, including:
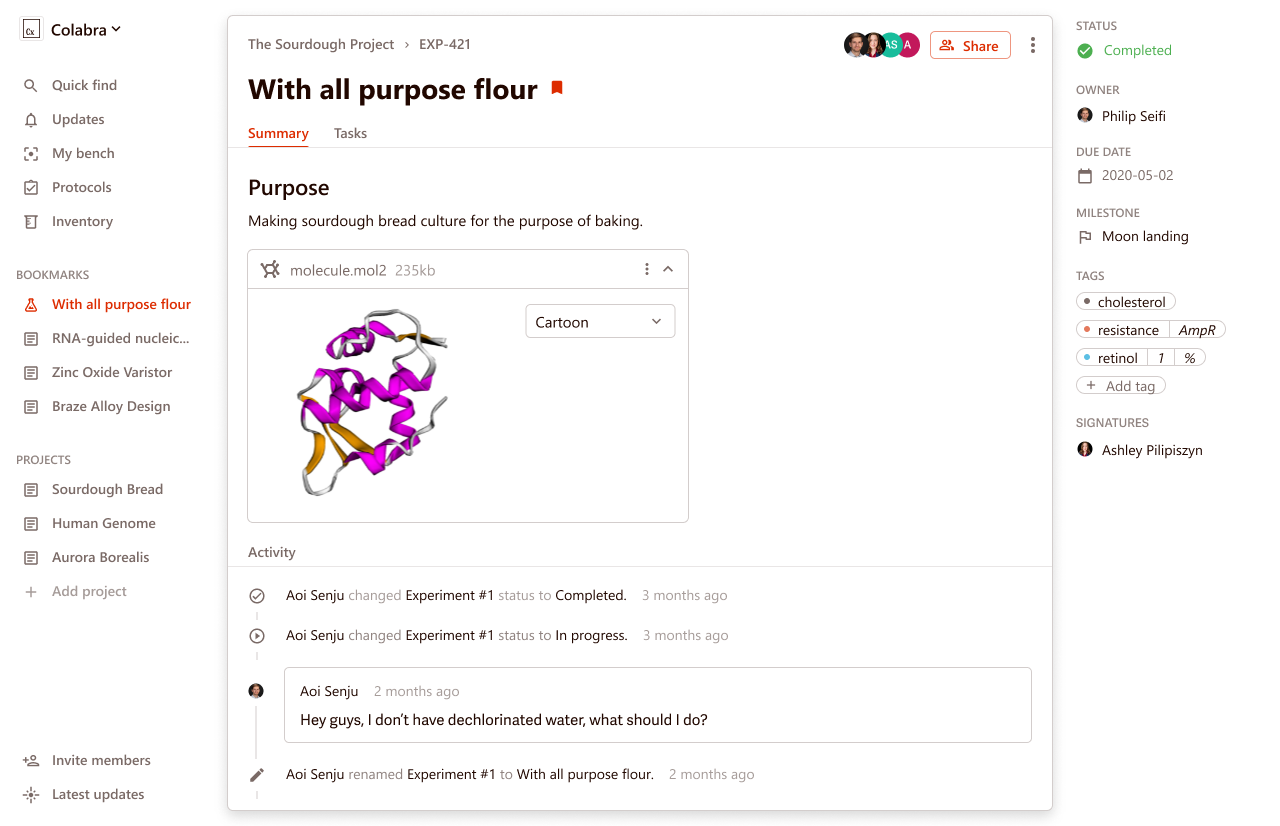
- An intuitive interface for improved usability and a high acceptance rate. Colabra offers a modern, easy-to-navigate interface.
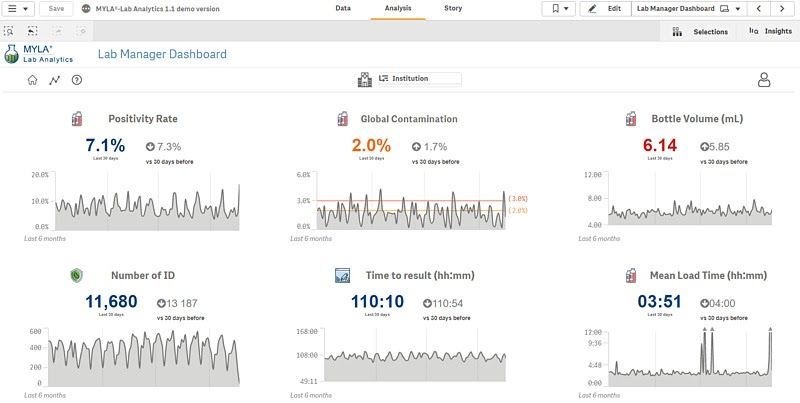
- Analytics features relevant to your specific objectives. MYLA Lab Analytics offers a variety of dashboards and modules designed specifically for microbiology laboratories.
- Sharing and collaboration tools that lab personnel can adopt easily. Colabra is an example of an ELN that puts a particular wemphasis on collaboration. It enables admins to create experiments and assign them to specific team members. It also allows for real time editing, as well as commenting directly within the lab notebook to document results and discussions in one place.
- Integrations with other laboratory software to make editing and sharing documents simpler. For example, Colabra easily integrates with Google Sheets, Miro, Deepnote, and more.
- Notification features that enable team members to receive updates in real-time.
- Built-in compliance tools or integration opportunities. Colabra offers compliance functionalities with FDA Title 21 CFR Part 11 for operating in a regulated GxP environment, SOC 2 Type 2 certified for data protection, and regional standards like GDPR, ePrivacy, CCPA, LGPD, and PECR.
- Protocol and SOP management features. Colabra has an easy-to-use protocol creation tool.
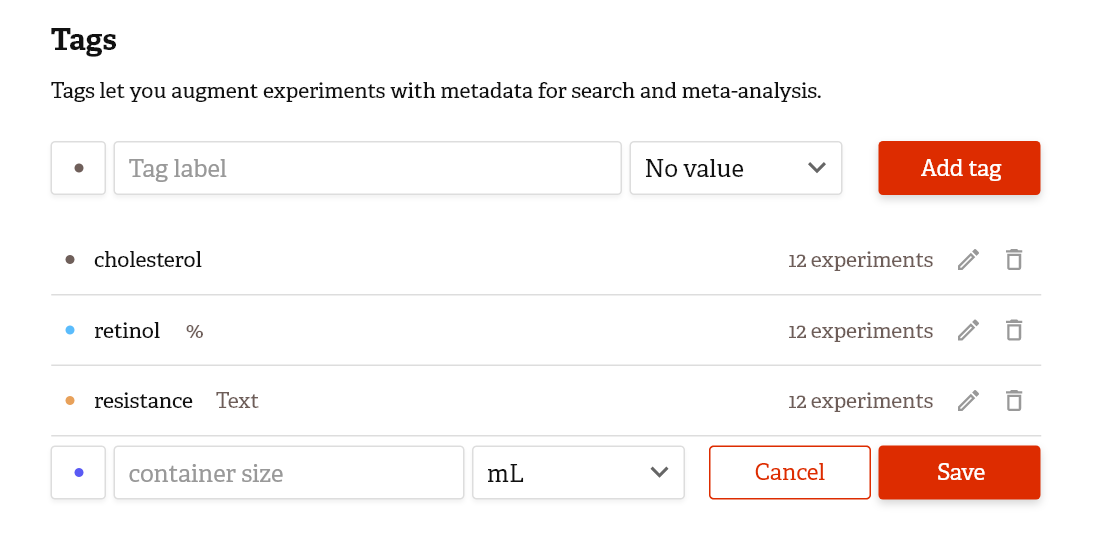
- Easy-to-use data organization systems such as intuitive filing systems and tag managers. For example, Colabra enables you to search for data based on author, tag, project category, and other criteria.
Legal compliance
The most relevant legal frameworks to be considered in your data management may be (but are not limited to) the following regulations.
FDA 21 CFR Part 11
Any electronic records maintained and stored according to Part 11 may be admissible in federal agency proceedings or federal legal proceedings (for example, with patent contestations).
In general, the FDA allows records to be stored and maintained in either electronic format or paper form. If a user elects to store and maintain records in an electronic format, then Part 11 requires that said electronic records include controls to ensure the electronic record’s authenticity, integrity, and confidentiality (when relevant).
This means an electronic record must:
- Be validated. Validation means the computer system utilized to maintain electronic records complies with all the requirements of Part 11, and the system has been tested to ensure said compliance.
- Generate accurate and complete records. The electronic records system must store, print, and transmit a complete and accurate copy of any electronic record stored in the system. A copy is accurate if the record is entered without alterations, deletions, or additions. The system must detect any invalid or altered records. And the system must retain the records for the applicable period.
- Have controlled access to electronic records. Each authorized user should be provided with an ID unique to them. Providing password protection for each authorized user would be a starting point. Additionally, automatically logging out an authorized user after a specified period of inactivity would reduce unauthorized access to electronic data.
- Create an audit trail. The system software must automatically generate an audit trail to ensure the records are accurate and have not been tampered with, including an audit trail to record the date and time of operator entries and any actions to create, modify, or delete records. The audit trail should list the date and local time of any entry to an electronic record, including the person making the entry. Any additions, deletions, or amendments to a previously entered record should not be overwritten. Instead, any changes to a previously-entered document should be reflected as a new document so that both documents can be viewed independently. The original and amended documents should be stored for a required retention period.
- Maintain digital signature standards. All digital signatures of an electronic record shall include the printed name of the person signing the record, the date and time when the signature was entered, and the “meaning” of the signature. The “meaning” of the signature is the relationship of the person signing the record to the electronic record, such as the author, the responsible party, the person reviewing the record, or the person approving the record. Electronic signatures entered on electronic records should be linked to the electronic record to prevent the electronic signature from being removed, copied, or transferred to another record by ordinary means.
A user should be educated and trained on the proper use of electronic records, and written policies should be established and implemented to ensure authenticity and integrity of records and to deter record and signature falsification.
Please keep in mind that this document does not provide detailed information or legal advice.
SOC 2 Type 2
System and Organization Controls (SOC) is a standard for security compliance for software companies. Its guidelines and policies help businesses protect customer data stored in the cloud.
SOC 2 fulfills five trust principles (security, availability, processing integrity, confidentiality, and integrity).
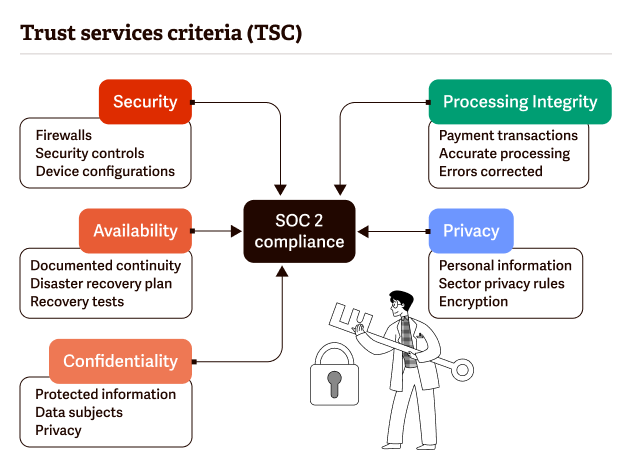
To get SOC 2 certified, organizations must undergo by outside auditors a thorough multi-month audit of their R&D cloud infrastructure, software, HR processes, and policies around handling customer data to ensure they have strict security procedures and controls in place.
For organizations to be SOC 2 Type 2 compliant, an independent auditor would review the following practices and policies:
- Infrastructure: the physical and hardware elements of a system.
- Software: the programs and operating software of a system.
- People: the personnel relevant to the operation of a system.
- Procedures: automated and manual procedures necessary for the operation of a system.
- Data: the information used and supported by a system.
SOC 2 certification should be considered mandatory for any DMS solutions.